Genomic Integrity Karyotyping Services 
Deep G-Banding Analysis to Assess Cell Batch Genomic Integrity.
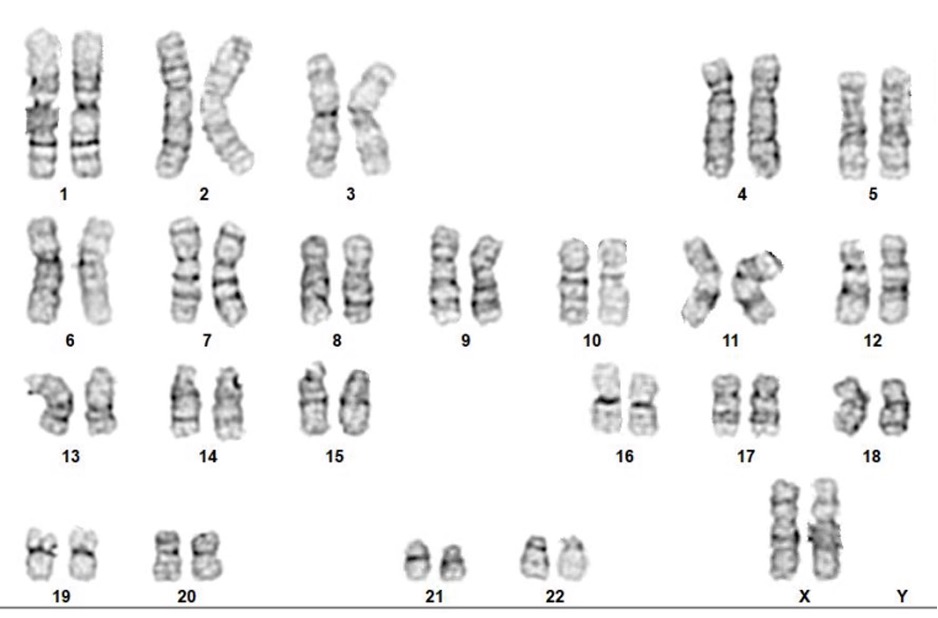
Above Fig.: G-banded karyogram of a genomic integrity baseline wild type T cell sample for comparison with edited T cells.
Develop a Study Plan with KromaTiD to Support Your Cell Therapy Development.
Genomic Integrity Service Features:
- Developed by KromaTiD to meet the unique needs of cell & gene therapy programs.
- Supports all stages of development from R&D to release of drug product.
- Research > Preclinical > Clinical Batch Release
- Detection of edit associated and other structural variants from 20-200+ cells per spread.
- Our Genomic integrity Karyotyping reports include detailed structural analysis and individual karyotype of each cell plus the overall karyotype for each sample. A genomic integrity comparison can also be provided highlighting the variances between unmodified donor cells and the cell and gene therapy drug product.
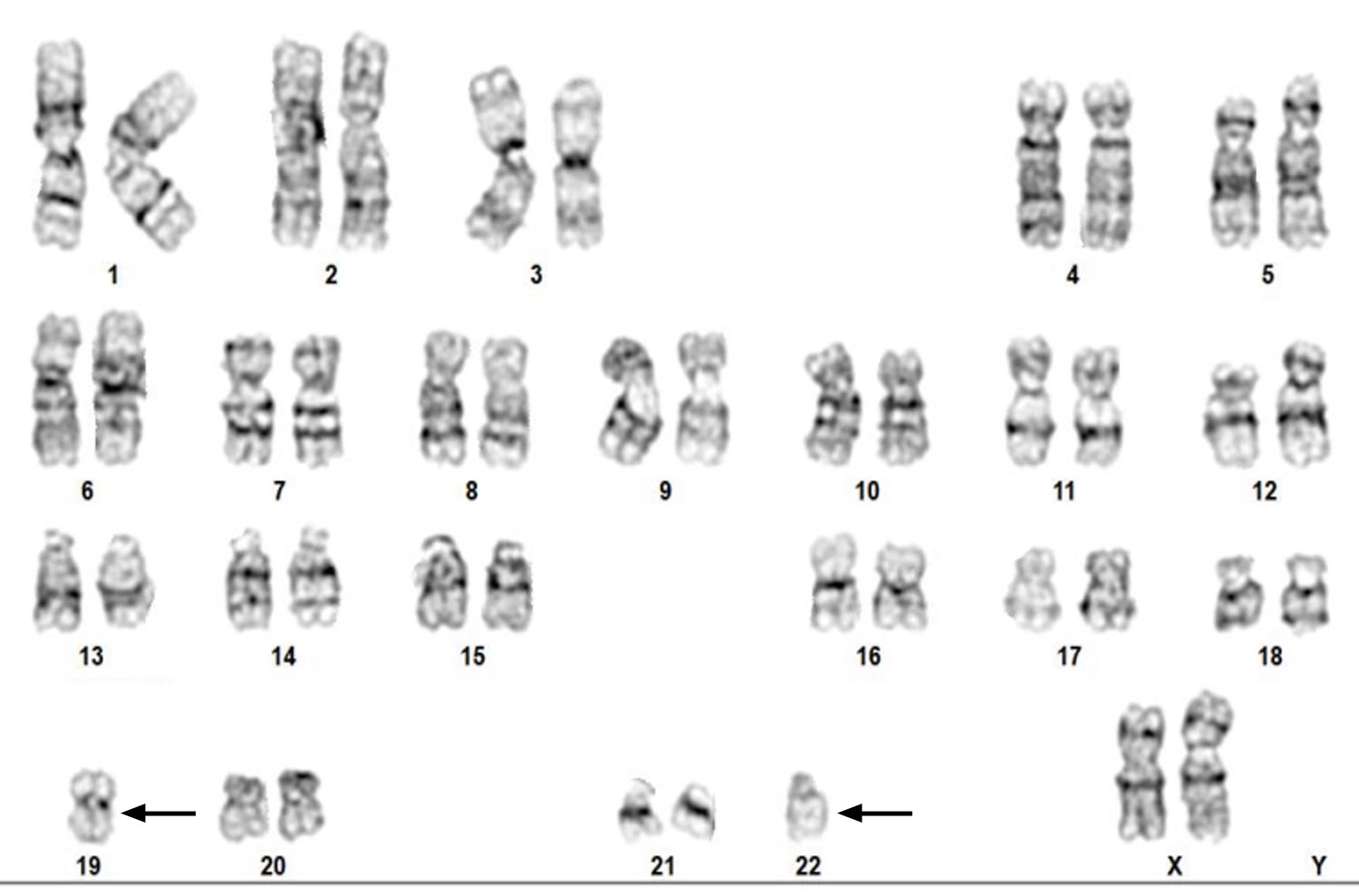
Above Fig.: G-banded karyogram of an edited T cell sample showing loss of one homolog each for chromosomes 19 and 22.
Distinguish between Origins of Genomic Abnormalities.
All cell handling and editing processes can cause structural variants, but determining which specific event or step is most likely to have triggered changes in the genome can be of tremendous value.
By comparing data from loci proximal to edit sites, it is possible to distinguish between different origins of genomic abnormalities, such as the editing process itself, culture expansion, random chance, or abnormalities which were pre-existing.
This insight can go a long way towards reducing the risk of unintended genomic changes, and of mitigating the impact of any which do occur.
KromaTiD Genomic Integrity reports provide a highly detailed analysis for every cell in each sample including enumeration of 28 types of chromosomal rearrangement events. See below for more information on our reporting options.

Above Fig.: Portion of sample genomic integrity report showing tallied abnormalities.
Expert, Detailed Reporting on Chromosomal Variants.
A G-Banding Genomic Integrity Report summarizing all the information necessary to interpret results is provided for all samples undergoing Genomic Integrity Karyotyping. This includes the overall karyotype outcome for each sample, as well as the karyotype designation for each cell and the number of abnormalities by type.
The G-Banding Genomic Integrity Pair-Wise Comparison Report can be added to the service and provides a side-by-side results comparison between a wild type control and gene-edited test sample. A Fisher’s exact test provides p-values for significance determination. Low-frequency aneuploidies, for example, can be assessed for statistical significance based on the variance from the baseline sample.
KromaTiD can qualify any of our karyotyping services against your drug product specifications and provide a customized, Genomic Integrity Drug Product Release Test to help accelerate your products to market.
*For GLP Pricing Please Inquire
*Complex Cell Type includes those, such as cancer cell lines, which are likely to have complex structural rearrangements, abnormal modal numbers, and potential instability creating highly variable karyotype from cell to cell.
