
Much of the biology-related news in recent decades has to do with scientific breakthroughs involving the very small. With the naked eye, we cannot see our own skin cells, yet we know there are scientists devoting their careers to the fight against skin cancers. Microscopic skin cells are the battlefield on which life-and-death combat is waged against a foe that would kill us. The same can be said about cells in the brain, the gut, the lungs, and cells in every other part of our bodies. Most people understand that microscopes can be used to see objects on this scale, but there are limits to what microscopes reveal by just making something appear bigger. A stick bug standing on a pile of twigs may remain just as well camouflaged even when viewed with a magnifying glass. Chemical stains can help distinguish specific types of objects within a sample, but they are a broad brush, perhaps staining all of a certain kind of molecule within a group of cells. They cannot, for instance, highlight the location of a single gene on a chromosome or one protein on a cell’s surface. A DNA molecule encoding a specific gene’s sequence can locate a matching (“complementary”) sequence within a cell’s nucleus, but this is not very useful on its own because that DNA will be invisible. It will be camouflaged among countless other organic molecules in the biochemical soup that is a cell’s cytoplasm. However, a scientist may attach a second, fluorescent molecule to that piece of DNA before sending it into the cell. Astronauts may spot a distant city at night by its electric lights. Biologists too can peer from on high, down the eyepieces of a microscope to spot the fluorescence of a molecular beacon, perhaps uncloaking an undiscovered Achilles’ heel of some cruel disease.
There exists a multitude of molecular fluorescent tags (“fluorochromes”) in use today. Many are derived from xanthene. The similar molecular structure hints at their common chemical origin.
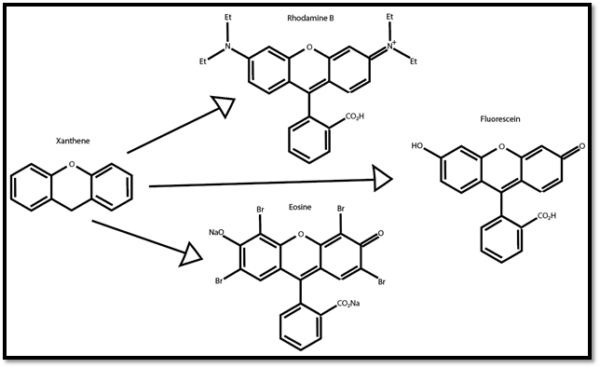
Figure 1: Xanthene and its derivative fluorochromes.
Another family of fluorochromes are cyanine derivatives, generally abbreviated as “Cy” followed by a number. For example, “Cy3,” “Cy5,” or “Cy5.5.”
Fluorochromes can be bound to DNA, RNA, proteins like antibodies, and many other types of molecules. The new fluorochrome-tagged molecule can then be used to probe a sample in search of a specific molecular target, be it a gene, a cell surface protein, or a target within the cytoplasm.
Each fluorochrome has an absorption peak, that is, light within a certain color (wavelength) range that will energize the molecule. It also has an emission peak, light within a certain color range that the molecule emits to release that energy. Whatever hardware is used to gather data from the probe, such as a fluorescence microscope or a flow cytometer, must be able to supply light the molecule can absorb and be able to detect the light given off by the molecule in turn.
NASA engineers send rovers to other planets to explore the solar system. Remotely operated submersibles are sent to search the lowest depths of the world’s oceans. So too within the fields of the biological sciences, fluorescently labeled molecular probes explore the kelp forests of DNA strands within cell nuclei, or the bobbing deserts of cell membranes where trans-membrane proteins surface and submerge like submarine periscopes. Those researchers that gather data via molecular probes shed light on the hidden messages within the molecules of life, advancing health and longevity through those tiny fluorescent lenses.
